Behind the Expansion: Inside Our Human Factors Research Facilities

The opening of the Crux Technology Centre marks an important step for our teams and the partners we work with across the healthcare sector. The new facility expands the specialist environments available to our engineers, designers and researchers developing the next generation of medical devices.
While the opening event offered a first glimpse of the centre, the full impact becomes clearer inside the environments where studies take place and products are refined.
Designed For Real World Use
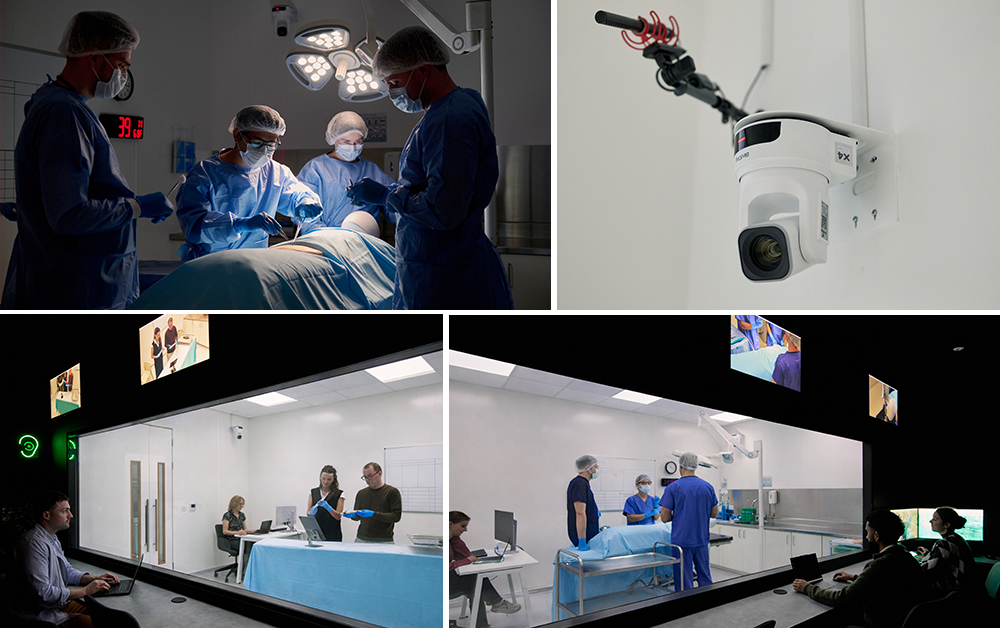
One of the most distinctive additions is the surgical Human Factors suite. This environment recreates clinical settings with a high level of realism, allowing teams to observe how devices are used in context.
Researchers can observe how clinicians use devices within simulated clinical workflows, helping teams assess handling, usability and workflow considerations. Adjacent viewing rooms allow clients and usability experts to observe sessions live, creating shared visibility into how devices perform in the hands of real users.
For clients, this means research insights can be captured earlier and with greater clarity. Decisions around ergonomics, usability and device handling can be informed by evidence gathered in environments that closely reflect real clinical practice.

Expanded Usability Research Capability
Alongside the surgical Human Factors suite, the Technology Centre strengthens Crux’s usability research capability across the full development journey.
Dedicated usability rooms support a wide range of studies, from early formative research exploring intuitive use and potential design refinements, to later-stage studies to evaluate the safety and effectiveness of the devices with the intended users.
These environments allow researchers to capture detailed feedback in a structured and consistent way, helping teams understand how devices are used and refine designs based on real-world insight.
A Connected Environment
The Technology Centre has been designed to bring research, engineering and design teams together throughout the life of a project.
Insights gathered during research sessions can move directly into design development and engineering evaluation. Prototype iterations can be refined quickly and returned to testing environments without delay. This connected workflow helps programmes maintain momentum while preserving the rigour required for healthcare products.
For partners developing complex medical technologies, this integrated approach supports faster cycles and more confident decision-making.
Greater Capacity for Complex Programmes
Crux now brings together more than 130 specialists across the UK and the US. The expanded facilities increase the scale and complexity of projects that can be supported while maintaining the close collaboration our clients value.
The result is a centre designed to support the increasingly sophisticated demands of modern healthcare innovation.
Experience the Technology Centre at HFES
Visitors attending the HFES International Symposium on Human Factors and Ergonomics, taking place in New York from March 22–25 2026, will also have the opportunity to explore the new facilities in an interactive way.
At the Crux booth, attendees can step inside the Technology Centre through a virtual reality experience. This immersive walkthrough offers a first-hand look at how Human Factors studies are conducted and how clinical environments are recreated.
If you are attending the symposium, we would be delighted to welcome you to the booth and show how the new Technology Centre is helping shape the future of healthcare technology.
Book a visit here.